|
 |
||||||||||
XAFS techniques
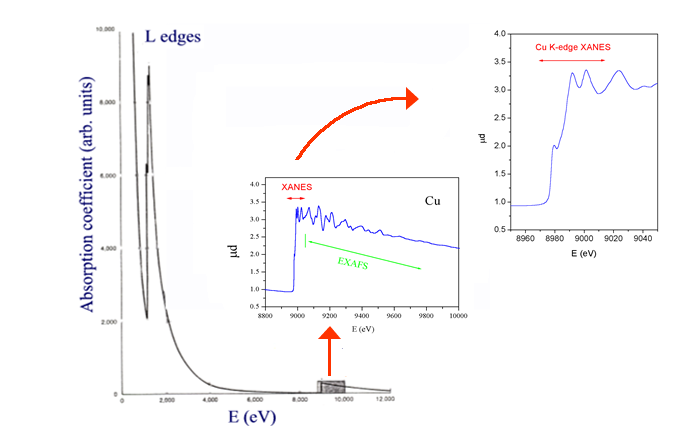
The prevailing rule of an x-ray absorption spectrum is the monotonic dependence of the x-ray absorption with the photon energy, interspersed with sharp edges (Fig. 4). However, the detailed shape of the edge and of the x-ray absorption spectrum above it contains useful structural information. In the dominant absorption process of photoeffect, an electron is ejected from the core, mostly from the deepest level accessible to the photon energy. The photoelectron is emitted in the form of a simple spherical wave when the atom is free, i.e. in a monatomic gas sample. If the atom is incorporated in dense matter, or even in a molecule, the outgoing wave and, consequently, the probability of the photoeffect, is modified by the surroundings. The problem of calculating the outgoing waves in the strong field of adjacent atoms in a solid or liquid sample is notoriously difficult: it has to be tackled in full for slow photoelectrons, i.e. when the incident photon energy is just above the threshold. This region of the absorption spectrum, so called XANES ( = X-ray Absorption Near-Edge Structure) contains valuable information on chemical bonds and the site symmetry [5,6].
|
|
|
Fig.
4. X-ray absorption coefficient of copper in the
region of L and K edges. The box area is expanded in the inset
to show EXAFS and XANES signal. |
Further out from the absorption edge, in the EXAFS ( = Extended X-ray Absorption Fine Structure) spectral region, the problem of the photoelectron wave is considerably simplified [6,7]. With the shorter photoelectron wavelength, the adjacent atoms scatter the photoelectron as point obstacles, each contributing a tiny wavelet (Fig. 5). The interference pattern of the photoelectron wavelets modifies the probability of the photoeffect. When the absorption spectrum is scanned by changing the photon energy, the energy of the photoelectron changes. Consequently, its wavelength varies, and the interference of the wavelets changes from constructive to destructive and back again. Each atom scatterer contributes a harmonic oscillatory mode, together they form a complex quasiperiodic EXAFS signal: Fourier analysis of the signal resolves the harmonic components into a probability vs distance diagram (Figs. 6, 7). Its peaks occur at rather accurate values of the neighbor atom distances. In addition, the coordination number and chemical species of the neighbor atoms, as well as the statistical spread of their distances due thermal motion or static disorder can be deduced from the size and shape of the peaks.
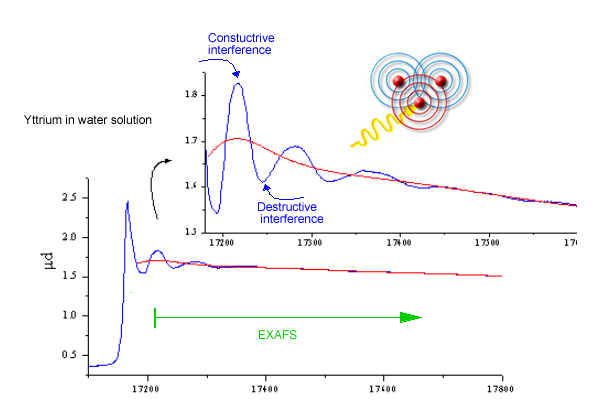 |
Fig.
5. Interference of photoelectron waves scattered
from neighbor atoms and the resulting oscillation in x-ray photoabsorption
probability. |
 |
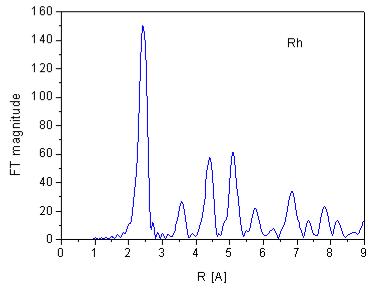 |
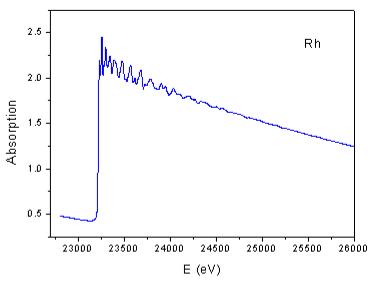
| Fig. 6. EXAFS signal of rhodium metal at 80 K above the K-edge (a) and its Fourier transform magnitude (b). | |
 |
 |
 |
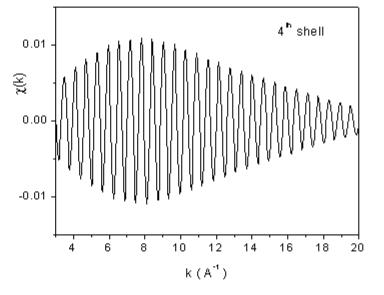 |
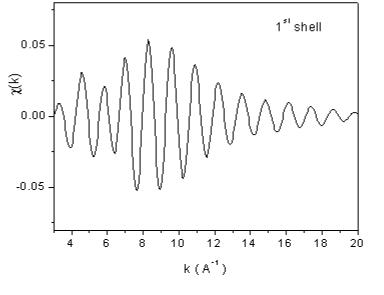
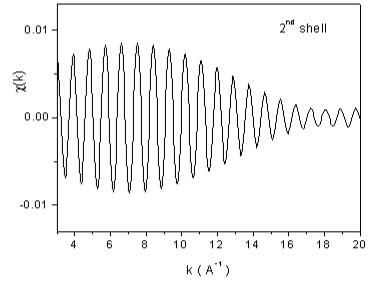
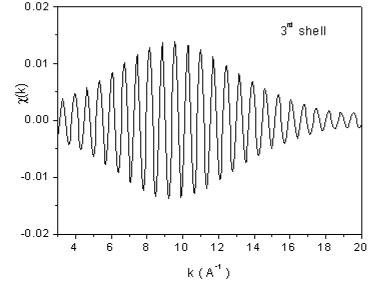
| Fig. 7. The contributions of the nearest four shells of neighbors to the Rh EXAFS signal from Fig 6, recovered from the largest peaks in the Fourier transform. | |
The basic EXAFS
experimental method is the standard absorption spectrometry of a thin
homogeneous sample of the investigated material above the absorption
edge of the element under study. Optimum thickness of the sample is
of the order of 10 microns for pure elements (metal foils) and accordingly
larger when other (lighter) elements are present. Powdered samples are
conveniently attached to layers of adhesive tape or mixed with a light
matrix and pressed into thin pellets. The sample must be at least locally
homogeneous: if not, the unavoidable small deviations of the x-ray beam
position on the sample are translated into spurious intensity variations
mimicking the EXAFS signal.
The sensitivity of the basic EXAFS experiment is not very high: the
measured element must contribute at least a few percent to the beam
absorption to produce a meaningful structural signal. Weaker signals
tend to be drowned in the statistical noise of the beam. In an alternative
method the fluorescence photons from the sample are monitored instead
of the transmitted beam, whereby the sensitivity is improved by one
or two orders of magnitude. In TEY ( = Total Electron Yield) detection
mode the emitted electrons from the sample are registered [8].
Another order of magnitude may be gained in sensitivity; the smaller
penetration
depth of the electrons, however, limits the sensitivity to a thin layer
at the surface of the sample. A good surface sensitivity is provided
also by measuring the intensity of the totally reflected x-ray beam:
the method requires a perfectly flat surface, though, which precludes
it for routine analysis [9]. A
specific variant of EXAFS, x-ray magnetic circular dichroism (XMCD)
has been developed for investigation of magnetic
materials [10,11].
The scan of a standard EXAFS spectrum requires about 20 minutes on a
synchrotron beamline and several hours on a conventional laboratory
x-ray generator. On modern synchrotron x-ray sources with high brilliance
much faster detection modes have been developed for studying chemical
reactions in real time (Quick EXAFS = QEXAFS 12). Currently, 100 ms
for a scan is available, with a promise of a hundredfold improvement
with the next generation of coherent x-ray sources (TESLA in Hamburg).
The range of the elements amenable to a routine EXAFS analysis depends
on the x-ray monochromator. The low-energy limit of the most widely
used Si(111) monochromators is around 3 keV, translating to Z = 16 (K
edge of sulfur). With special monochromators, the technique can be extended
to about 2 keV (K edge of aluminium). For the lightest elements (C,
O, N) diffraction gratings are used instead of crystals for beam monochromatization:
in this low-energy region (200 - 600 eV), the typical span of the EXAFS
signal (1 keV) requires or exceeds the entire monochromator range, so
that only XANES spectra are studied [5].
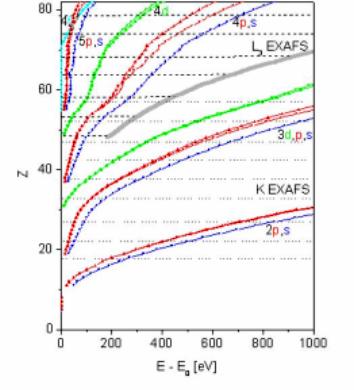 |
At the high-energy end, the limit is defined by the large-scale parameters of the x-ray source: the storage-ring energy and radius define the critical photon energy above which the intensity of the synchrotron radiation decreases exponentially [4]. Consequently, L3 edge EXAFS instead of K edge is exploited for heavier elements. The switch to L3 is not recommended for elements below antimony (L3 edge 4132 eV, K edge 30491 eV), since the range of the L3 EXAFS signal, cut off by the subsequent L2 edge, is too short so that the spatial resolution of the method is seriously impaired. In the schematic overview of accessible EXAFS intervals in Fig. 8, the relative position of the limiting L2 edge is shown by the thick gray line. However, modern insertion devices (wigglers) generate the high-energy photons much more abundantly and the switch to the L3 EXAFS is practised at higher energies (and atomic numbers) depending on the monochromator resolution. Mostly, L3 edge is exploited for lanthanides and heavier elements. |
Fig. 8. The useful interval of EXAFS signals (horizontal dotted lines) as a function of the atomic number. With K-edge energy out of the range of standard monochromators, L3 edge EXAFS is measured for heavier elements. The thick gray line shows the relative position of L2 edge truncating the L3 EXAFS signal. The relative excitation energies of sharp multielectron photoexcitation features in the EXAFS interval are shown by dots. |
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
E-mail:iztok.arcon@p-ng.si Last change: 31-Mar-2004 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||